
The time to first admission did not show a treatment effect ( Figure 5) although uncertainty in this endpoint was high. Numbers of overall hospitalizations (SAEs Table 2) were less in the canakinumab arm compared with placebo arm ( Table 2), resulting in a mean SAE rate of 0.9 per patient vs 1.1, respectively. Nearly all study hospitalizations, recorded as SAEs, resulted from SCA-related pain crises or complications thereof. Correspondingly, median hsCRP baseline values also trended higher in the canakinumab group. Compared with the placebo group at baseline, patients assigned to the canakinumab group trended toward higher disease activity during the screening period, as reflected by trends toward lower hemoglobin levels and higher daily levels of pain and fatigue and opiate/nonopiate analgesic use ( Table 1). All patients, except for 1 placebo arm patient, were receiving HU background therapy of comparable median daily dose levels between the 2 study arms. There were no AE- or drug-related discontinuations. Two patients discontinued in the canakinumab arm due to subject/guardian decision, and 5 discontinued in the placebo arm due to either decision by patient/guardian (n = 1) or physician (n = 2) or were lost to follow-up (n = 2). Of 25 patients randomized to canakinumab and 24 to placebo, data for the 24-week blinded period are available for 23 and 19 patients, respectively (supplementary Data). Overall, baseline demographics were well matched for age, BMI, gender, and ethnicity across the 2 treatment groups ( Table 1). Protocol and consent forms were approved by local ethics committee at each participating trial site. All participants ≥18 years provided written informed consent before study participation, with parent or legal guardian written informed consent required along with child’s assent for participants <18 years. Participants receiving regular RBC transfusion therapy or having received a transfusion in the past 3 months prior to screening were excluded. Participants receiving HU were eligible if stable dosing ≥60 days prior to screening. In addition, study participants were required at screening to have hsCRP >1.0 mg/L and detectable background pain as recorded by electronic patient-reported outcome device (eDiary defined as either average daily pain score >1 (see section on Endpoints) without analgesic use over a period of ≥7 days or ≥1 episode of pain requiring analgesia within a 14-day period. 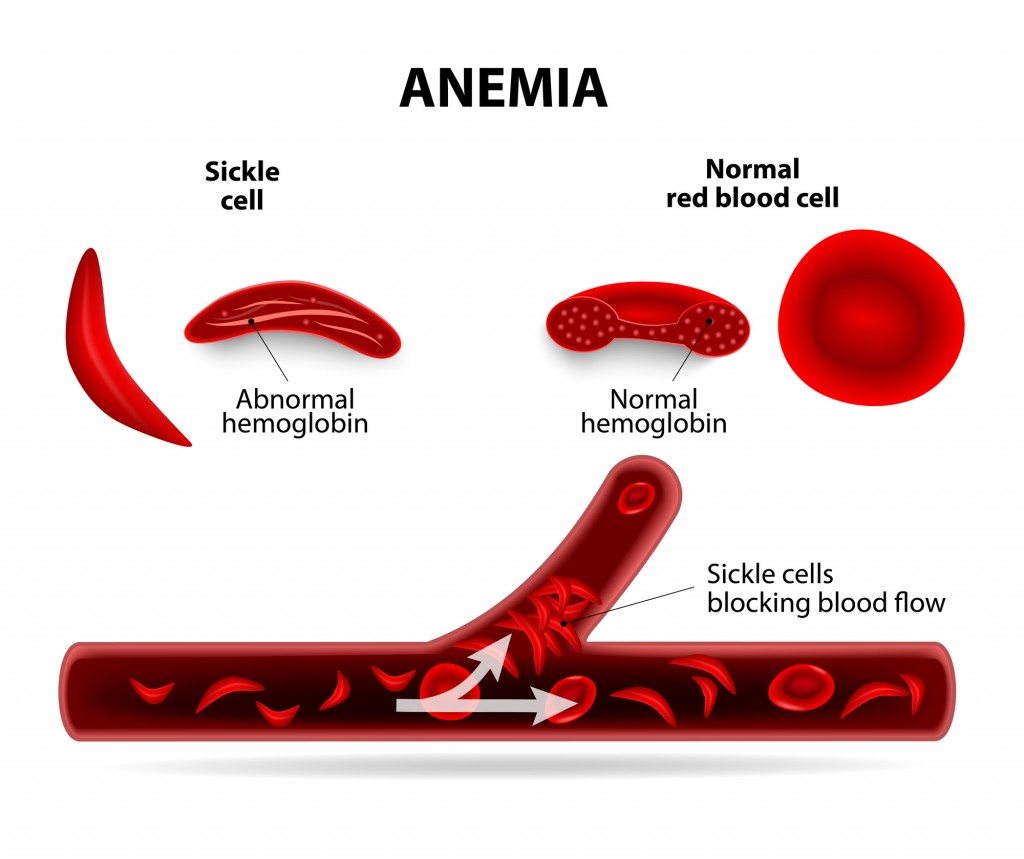
The study population included participants 8 to 20 years of age with homozygous HbSS or HbSβ 0-thalassemia who experienced in the year prior screening at least 2 vaso-occlusive pain episodes, defined as pain without non–sickle cell identifiable cause, requiring analgesia, and interfering with participants’ normal daily routine. This trial was registered at as #NCT02961218.Ī younger patient population was recruited to reduce the presence of irreversible tissue damage (eg, degenerative joint disease, stroke) and thus was more likely to show clinical improvement in response to targeted antiinflammatory treatment. 
These findings demonstrate that the inflammation associated with SCA can be reduced by selective IL-1β blockade by canakinumab with potential for therapeutic benefits. Canakinumab was well tolerated with no treatment-related SAEs and no new safety signal. Post hoc analysis revealed treatment effects on weight, restricted to pediatric patients. Although the primary objective (prespecified reduction of pain) was not met, compared with patients in the placebo arm, patients treated with canakinumab had reductions in markers of inflammation, occurrence of SCA-related AEs and SAEs, and number and duration of hospitalizations as well as trends for improvement in pain intensity, fatigue, and absences from school or work. All but 1 of the 49 enrolled patients were receiving stable background hydroxyurea therapy. Measured outcomes at baseline and weeks 4, 8, 12, 16, 20, and 24 included electronic patient-reported outcomes, hospitalization rate, and adverse events (AEs) and serious AEs (SAEs). In this randomized, double-blind, multicenter phase 2a study, patients aged 8 to 20 years with SCA (HbSS or HbSβ 0-thalassemia), history of acute pain episodes, and elevated high-sensitivity C-reactive protein >1.0 mg/L at screening were randomized 1:1 to received 6 monthly treatments with 300 mg subcutaneous canakinumab or placebo. We hypothesized that IL-1β blockade by canakinumab in patients with SCA would reduce markers of inflammation and clinical disease activity. Excessive intravascular release of lysed cellular contents from damaged red blood cells (RBCs) in patients with sickle cell anemia (SCA) can activate the inflammasome, a multiprotein oligomer promoting maturation and secretion of proinflammatory cytokines, including interleukin-1β (IL-1β).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
